See how simple it is to apply SANTYL Ointment
Step-by-step application
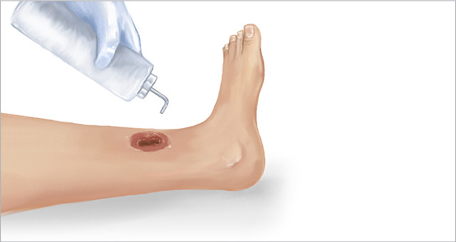
1. Moisture balance
- Wounds with sufficient moisture will naturally activate collagenase enzyme
- Wounds heal better in a moist environment
- Nerve endings are protected by moisture24
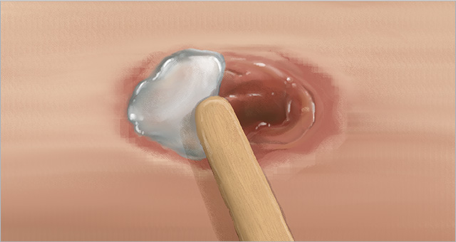
2. Edge to edge
- Apply SANTYL Ointment out to the edges of the wound
- New epithelial and granulation tissue grows from the wound’s edges
- Take care not to extend beyond the wound surface although it does not harm healthy tissue
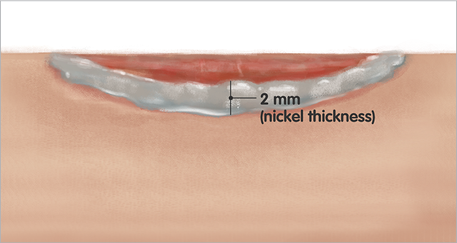
3. Nickel thickness
- Apply once daily at 2mm thickness or about the thickness of a nickel
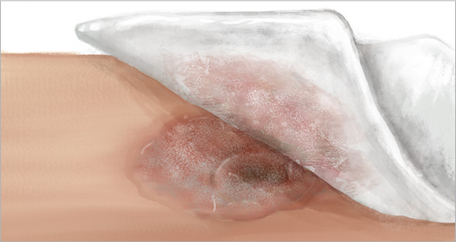
4. Daily dressing change
- Daily dressing changes help remove necrotic burden from wound area to help advance the healing process4
- Moisturize cover dressing with saline if wound is dry and there is no sufficient exudate
- The enzymatic action of SANTYL Ointment may be decreased by dressings containing iodine (I2) and some, but not all, dressings containing silver (Ag)
Compatibility with other products
![]()
SANTYL Ointment has been tested with cleansers, antimicrobial formulations, and silver dressings21
SANTYL Ointment is compatible with:
- Products that contain alkali metal salts (ie, sodium [Na] or potassium [K]) and earth alkaline metal salts (ie, calcium [Ca] or magnesium [Mg])
- Large non-ionic compounds and non-charged surfactant molecules
- Antimicrobial formulations
- Wound cleansers containing large block co-polymer surfactants (eg, poloxamers) or formulas with dilute sodium hypochlorite
The enzymatic action of SANTYL Ointment is decreased by:
- Dressings containing iodine (I2) and some, but not all, dressings containing silver (Ag)
- Wound cleansers designated to remove impurities, bacteria, and debris
- Antifungal agents and products containing EDTA
For best results:
When using antibiotic powder for infection, apply prior to SANTYL Ointment. If infection persists, discontinue SANTYL Ointment until infection is controlled.
Silver dressings |
Aquacel™ Ag |
|
|
ALLEVYN♢ Ag |
||
Iodine dressings |
IODOFORM™ |
|
Various other dressings |
Hydrofera™ |
|
|
PROSHIELD♢ |
||
|
Anasept® Spray |
||
Wound cleansers |
Microcyn™ Rx Spray |
|
|
¼-strength Dakin's solution |
||
|
Mafenide Acetate |
||
Antimicrobials |
Mupirocin |
Mode of Action references
- Loveluck J, et al. ePlasty. 2016;16:183-195.
- Galiano RD, et al. PlastReconstr Surg Glob Open. 2018;6(1):e1560.
- Pellino G, et al. Surg Innov. 2014;21(2):204–212.
- Kilpadi DV, et al. Wound Repair Regen. 2011;19(5):588-596.
- Malmsjö M, et al. ePlasty. 2014;14:1-15.
- Ma Z, et al. Exp Ther Med. 2016;11(4):1307-1317.
- Xia CY, et al. Mol Med Rep. 2014;9(5):1749-1754.
- Birke-Sorensen H, et al.. Journal of plastic, reconstructive and aesthetic surgery: JPRAS 64 Suppl, S1–S16 (2011)
- Scalise A, et al. Int Wound J. 2016;13:1260–1281.
- Shim HS, et al. Biomed Res Int. 2018;2018
- R. G, R. D, J. S, et al. The effects of a single use canister-free Negative Pressure Wound Therapy (NPWT) System* on the prevention of post surgical wound complications in patients undergoing bilateral breast reduction surgery. Paperpresented at: The British Association of Aesthetic Plastic Surgeons (BAAP's) 30th Annual Scientific Meeting; 2014; London.
- Hudson DA, Adams KG, Van Huyssteen A, Martin R, Huddleston EM. Simplified negative pressure wound therapy: clinical evaluation of an ultraportable, no-canister system. Int Wound J. 2015;12(2):195-201.
- Kirsner R, Dove C, Reyzelman A, Vayser D, Jaimes H. A Prospective, Randomised, Controlled Clinical Trial on the Efficacy of a single-use Negative Pressure Wound Therapy System, compared to Traditional Negative Pressure Wound Therapy in the Treatment of Chronic Ulcers of the Lower Extremities. Wound Repair Regen. 2019;27(5):519 - 529.
- Smith+Nephew 2018.Summary of rountine QA testing on MVP of PICO dressings. 2018. Internal Report. DS/18/153/R.
- Smith+Nephew 2019.Use of Moisture Vapour Permeability* (MVP) and Moisture Vapour Transmission rate** (MVTR) data to support product claims referring to moist wound healing. Internal Report. EO.AWM.PCSgen.001.v2.
- Saxena V, et al. Plast Reconstr Surg. 114:1086–96.
- Borquist O, et al. Annals of Plastic Surgery. 64: 789–93.
- Ichioka S, et al. Wound Rep Regen. 16(3); 460–465.
PICO active incision management references
- Malmsjö M, Huddleston E, Martin R. Biological Effects of a Disposable, Canisterless Negative Pressure Wound Therapy System. ePlasty. 2014;14:1 - 15
- Hudson DA, Adams KG, Van Huyssteen A, Martin R, Huddleston EM. Simplified negative pressure wound therapy: clinical evaluation of an ultraportable, nocanister system. Int Wound J. 2015;12(2):195-201.
- Payne C, Edwards D. Application of the Single Use Negative Pressure Wound Therapy Device (PICO) on a Heterogeneous Group of Surgical and Traumatic Wounds. ePlasty. 2014:152-166.
- Stryja J, Staffa R, Říha D, Stryjová K, Nicielniková K. Cost-effectiveness of negative pressure wound therapy in outpatient setting. Prolekare. 2015;94(8):322 - 328.
- Smith+Nephew November 2018.The Review Of Evidence Supporting The Use Of PICO In Wounds ≥2cm In Depth. Internal Report. EO.AWM.PCS230.001.v2.
- Casey C. Consistent delivery of therapeutic negative pressure levels by a singleuse negative pressure wound therapy system (sNPWT)* in a wound model. Paper presented at: EWMA; 2019; Gothenburg, Sweden
- Smith+Nephew January 2019.Air Leak Tolerance Report: A comparison of PICO v2 (PICO 7 and PICO 14) Devices to PICO vl.6 (PlCO) Devices. Internal Report. RD/19/006.
- Smith+Nephew December 2018.PICO 14 Service Life Testing: 14 Day Device Lifespan. Internal Report. RD/18/132.
- Smith+Nephew 2021.PICO™ Pressure Mapping Study. Internal Report. DS/19/211/R - Part B.
- Smith+Nephew July 2018.PICO 7Y Non-NPWT Wound Model Summary. Internal Report. DS.18.260.R.
- Smith+Nephew 2020.Bacterial barrier testing of the PICO dressing. Internal Report. 2001002.
- Smith+Nephew 2018.Summary of rountine QA testing on MVP of PICO dressings. 2018. Internal Report. DS/18/153/R.
- Gilchrist B, Robinson M, Jaimes H. Performance, safety, and efficacy of a single use negative pressure wound therapy system for surgically closed incision sites and skin grafts: A prospective multi-centre follow-up study. Paper presented at: SAWC; 2020; Virtual
- Sharpe A, Myers D, Searle R. Using single use negative pressure wound therapy for patients with complicated diabetic foot ulcers: an economic perspective. Wounds UK. 2018;14(3):80-137.
- Myers D, Sharpe A. EP549 Service Delivery in Complex DFU Patients using Single Use NPWT - A UK Perspective. Paper presented at: EWMA; 2018; Krakow.
- Hurd T, Gilchrist B. Single use negative pressure wound therapy (sNPWT) in the community management of chronic open wounds deeper than 2cm. Paper presented at: Symposium on Advanced Wound Care/Wound Healing Society Meeting; 2020; Abu Dhabi.
- Kirsner R, Dove C, Reyzelman A, Vayser D, Jaimes H. A Prospective, Randomised, Controlled Clinical Trial on the Efficacy of a single-use Negative Pressure Wound Therapy System, compared to Traditional Negative Pressure Wound Therapy in the Treatment of Chronic Ulcers of the Lower Extremities. Wound Repair Regen. 2019;27(5):519 - 529.
- Wang E, Tang R, Walsh N, et al. Topical negative pressure therapy and compression in the management of venous leg ulcers: a pilot study. Wound Practice and Research 2017;25(1):36-40.
- Smith+Nephew 2020.Negative Pressure Wound Therapy and Pressure Garment Therapy. Internal Report. EO.AWM.PCS261.002.v2.
General references
- Kirsner R,et al Wound Repair Regen. 2019,27(5),519 - 529
- Kirsner RS. et al. Wound Manag & Prev. 2020:66(3):30-36.
- Dowsett C, E’t al. wounds lnternat,onal 2017;8(2):52-58.
- Guest Jf, et al. J Wound Care. 2017;26(6):292-303.
- Fletcher J. et at Wounds UK. 2022. Available at, http://www.pcdsociety.org/resources/details/active-treatment-non-healing-wounds-community
- Nursery research - TSO
- UK survey - citalton TBD
- Smith+Nephew 2018. Internal Report. OS 18.260.R.
- Hurd T. et al. Ostomy Wound Manage 2014;60(3):30-36
- Gilchrist B. et al Performance, safety, and efficacy of a single use negative pressure wound therapy system for surgically closed incision sites and skin grafts: A prospective multi-centre follow-up study. Paper presented at: SWC; 2020.
- Smith+Nephew 2018. Internal Report. RD/18/137.
- Smith+Nephew March 2018. Internal Report, DS.18.066.R.
- Stryja J, et al Prolekare. 2015;94(8):322 - 328.
- Wang E. et al Wound Practice and Research 2017;25(1):36-40.
- Smith+Nephew 2020. lntemal Report. EO.AWM.PC5261.002.v2.
- Smith+Nephew 2018. Internal Report. EO.AWM.PCS230 001.v2.
- Smith+Nephew 2015. Internal Report. ST865 CT09/02.
- Hudson DA., et al. Int Wound J 2015;12(2):195-201
- Payne C., et al. ePlasty. 2014:152-166.
- Casey C., Consistent delivery of therapeutic negative pressure levels by a single use negative pressure wound therapy system (sNPWT)• In a wound model. Paper presented at: EWMA; 2019; Gothenburg, Sweden.
- Smith+Nephew 2019.P ICO Biomechanical Study. Internal Report. DS/19/211/R.
- Malmsjö. et al. ePlasty. 2014;14: l-15.
- Smith+Nephew 2020. Internal Report, 2001002
- Kilpadi DV, et al. Wound Repair Regen. 2011;19(5):588-596.
- Ma Z., et al E,p Ther Med 2016;11(4):1307-1317
- Xia CV., et al Mot Med Rep. 2014;9(5):1749-1754.
- Brownhill VR., et al. Adv Wound Care (New Rochelle) 2020;0(0): l-12.
- Patel A., et al. Comparison of wound closure in chronic lower extremity ulcers between single use negative pressure wound therapy and traditional negative pressure wound therapy: a real world analysis. Paper presented at: National Wound Conference; 2019: Las Vegas, NV, USA
- Hurd T., Gilchrist B., Single use negative pressure wound therapy (sNPWT) in the community management of chronic open wounds deeper than 2cm. Paper presented at: Symposium on Advanced Wound Care/Wound Healing Society Meeting: 2020; Abu Dhabi
- Mcmanus H., et al Bacterial retention within a multi-layered absorbent AIRLOCK’” Technology Single Use Negative Pressure Wound Therapy (sNPWT) dressing. Paper presented at EWMA: 2018; Krakow, Poland,
- Smith+Nephew. 2018. Internal Report. C5DAWM.24.056.
- Smith+Nephew. Internal Report. RD/19/006
- Smith+Nephew. Internal Report RD/18/132.
- Smith+Nephew 2018. Internal Report. DS/18/219/R V2
- Schwartz JA, et al. J Wound care. 2015;24(2).
- Smith+Nephew 2020. Internal Report, EO.AWM.PCS26l 002.v2
- Smith,Nephew 2016. Internal Report. DS.16.179.R.
- Smith+Nephew 2016. Internal Report DS.16.174.A.
- Smith+Nephew 2008. Internal Report. 05/08/062/Rl
- Smith+Nephew 2009. Internal Report DS/08/078/R2
- Smith+Nephew 2008. Internal Report. 05/08/062/R2
- Aicher B, et al. Journal of Vascular Nursing. 2017 Sep 1;35(3):146–56.

