Remove the barriers to healing.
The only FDA-approved enzymatic debridement agent indicated for debriding both chronic dermal ulcers and severely burned areas. Its unique mechanism of action selectively removes necrotic tissue without harming healthy tissue.
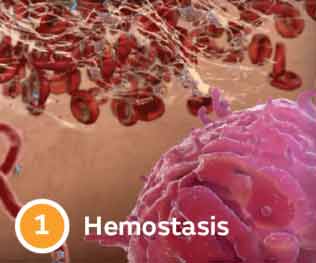
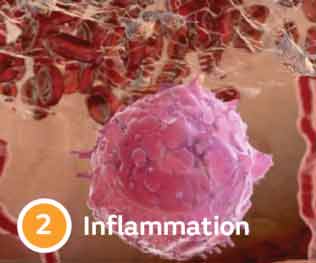
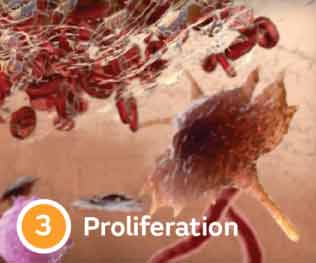
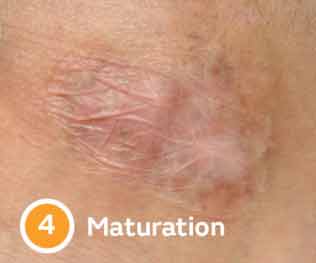
Four stages of wound healing
Delays keep wounds chronic
Wound chronicity can result from an imbalance in enzyme activity, resulting from comorbidities such as cardiovascular disease & diabetes.
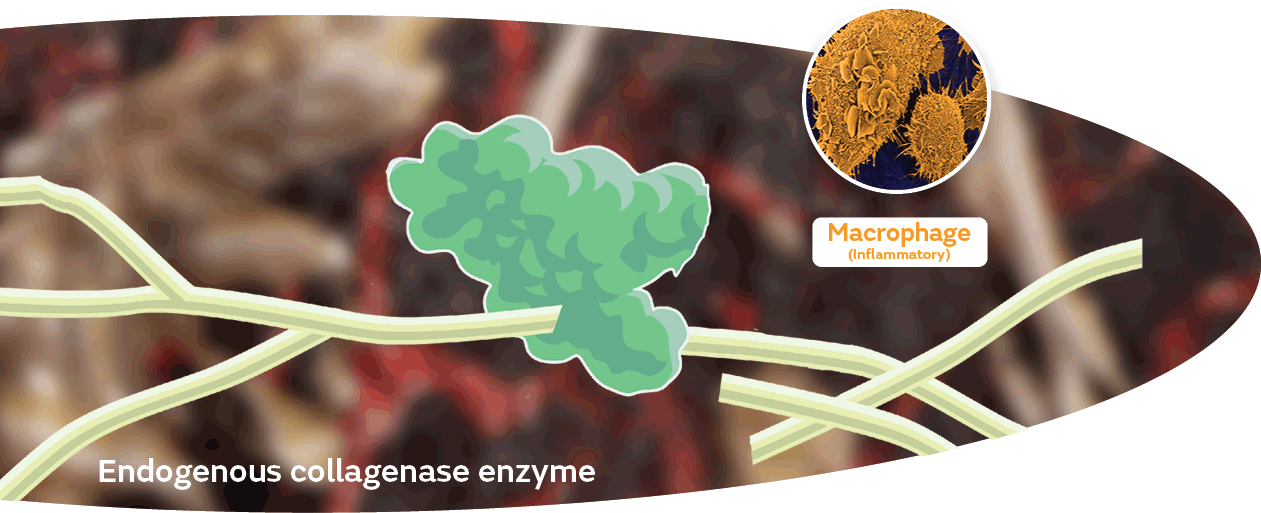
SANTYL Ointment prepares the wound bed at the microscopic level
The uniquely active and selective enzyme cleaves collagen molecules at seven sites, simultaneously clearing necrotic tissue and generating collagen polypeptide fragments.1,10,11
These bioactive peptides:5,10,11
- Break the cycle of inflammation12-14
- Recruit cells that lead to new tissue formation and blood vessel networks1,9
Watch SANTYL's mode of action
Build from the ground up with SANTYL Ointment
SANTYL Ointement has a wealth of established data to support its clinical efficacy
Mode of Action references
- Loveluck J, et al. ePlasty. 2016;16:183-195.
- Galiano RD, et al. PlastReconstr Surg Glob Open. 2018;6(1):e1560.
- Pellino G, et al. Surg Innov. 2014;21(2):204–212.
- Kilpadi DV, et al. Wound Repair Regen. 2011;19(5):588-596.
- Malmsjö M, et al. ePlasty. 2014;14:1-15.
- Ma Z, et al. Exp Ther Med. 2016;11(4):1307-1317.
- Xia CY, et al. Mol Med Rep. 2014;9(5):1749-1754.
- Birke-Sorensen H, et al.. Journal of plastic, reconstructive and aesthetic surgery: JPRAS 64 Suppl, S1–S16 (2011)
- Scalise A, et al. Int Wound J. 2016;13:1260–1281.
- Shim HS, et al. Biomed Res Int. 2018;2018
- R. G, R. D, J. S, et al. The effects of a single use canister-free Negative Pressure Wound Therapy (NPWT) System* on the prevention of post surgical wound complications in patients undergoing bilateral breast reduction surgery. Paperpresented at: The British Association of Aesthetic Plastic Surgeons (BAAP's) 30th Annual Scientific Meeting; 2014; London.
- Hudson DA, Adams KG, Van Huyssteen A, Martin R, Huddleston EM. Simplified negative pressure wound therapy: clinical evaluation of an ultraportable, no-canister system. Int Wound J. 2015;12(2):195-201.
- Kirsner R, Dove C, Reyzelman A, Vayser D, Jaimes H. A Prospective, Randomised, Controlled Clinical Trial on the Efficacy of a single-use Negative Pressure Wound Therapy System, compared to Traditional Negative Pressure Wound Therapy in the Treatment of Chronic Ulcers of the Lower Extremities. Wound Repair Regen. 2019;27(5):519 - 529.
- Smith+Nephew 2018.Summary of rountine QA testing on MVP of PICO dressings. 2018. Internal Report. DS/18/153/R.
- Smith+Nephew 2019.Use of Moisture Vapour Permeability* (MVP) and Moisture Vapour Transmission rate** (MVTR) data to support product claims referring to moist wound healing. Internal Report. EO.AWM.PCSgen.001.v2.
PICO active incision management references
- Malmsjö M, Huddleston E, Martin R. Biological Effects of a Disposable, Canisterless Negative Pressure Wound Therapy System. ePlasty. 2014;14:1 - 15
- Hudson DA, Adams KG, Van Huyssteen A, Martin R, Huddleston EM. Simplified negative pressure wound therapy: clinical evaluation of an ultraportable, nocanister system. Int Wound J. 2015;12(2):195-201.
- Payne C, Edwards D. Application of the Single Use Negative Pressure Wound Therapy Device (PICO) on a Heterogeneous Group of Surgical and Traumatic Wounds. ePlasty. 2014:152-166.
- Stryja J, Staffa R, Říha D, Stryjová K, Nicielniková K. Cost-effectiveness of negative pressure wound therapy in outpatient setting. Prolekare. 2015;94(8):322 - 328.
- Smith+Nephew November 2018.The Review Of Evidence Supporting The Use Of PICO In Wounds ≥2cm In Depth. Internal Report. EO.AWM.PCS230.001.v2.
- Casey C. Consistent delivery of therapeutic negative pressure levels by a singleuse negative pressure wound therapy system (sNPWT)* in a wound model. Paper presented at: EWMA; 2019; Gothenburg, Sweden
- Smith+Nephew January 2019.Air Leak Tolerance Report: A comparison of PICO v2 (PICO 7 and PICO 14) Devices to PICO vl.6 (PlCO) Devices. Internal Report. RD/19/006.
- Smith+Nephew December 2018.PICO 14 Service Life Testing: 14 Day Device Lifespan. Internal Report. RD/18/132.
- Smith+Nephew 2021.PICO™ Pressure Mapping Study. Internal Report. DS/19/211/R - Part B.
- Smith+Nephew July 2018.PICO 7Y Non-NPWT Wound Model Summary. Internal Report. DS.18.260.R.
- Smith+Nephew 2020.Bacterial barrier testing of the PICO dressing. Internal Report. 2001002.
- Smith+Nephew 2018.Summary of rountine QA testing on MVP of PICO dressings. 2018. Internal Report. DS/18/153/R.
- Gilchrist B, Robinson M, Jaimes H. Performance, safety, and efficacy of a single use negative pressure wound therapy system for surgically closed incision sites and skin grafts: A prospective multi-centre follow-up study. Paper presented at: SAWC; 2020; Virtual
- Sharpe A, Myers D, Searle R. Using single use negative pressure wound therapy for patients with complicated diabetic foot ulcers: an economic perspective. Wounds UK. 2018;14(3):80-137.
- Myers D, Sharpe A. EP549 Service Delivery in Complex DFU Patients using Single Use NPWT - A UK Perspective. Paper presented at: EWMA; 2018; Krakow.
General references
- Najjar PA, et al. Surg Clin N Am 2015;95(2):269-283. World Union of Wound Healing Societies (WUWHS). Consensus Document: Closed surgical incision management: Understanding the role of NPWT. https://woundsinternational.com/world-union-resources/closed-surgical-incision-managementunderstanding-the-role-of-npwt/.
- Merkow, R, et al. JAMA, February 3, 2015, Volume 313, Number 5.
- Nussbaum SR, et al. An. Value Health. 2018 Jan;21(1):27-32.
- Olsen et al. J Am Coll Surg. 2008 Sep;207(3):326-35.
- Olsen MA, et al. Archives of Surgery. 2008 Jan 1;143(1):53-60.
- Cotogni P, et al. J Crit Care Med 2015;4(4): 265-273.
- Jenks PJ, et al. Journal of Hospital Infection, 2014; 86(1), pp.24-33.
- Tanner J, et al. Journal of Hospital Infection. 2009 Jul 1;72(3):243–50.
- Aicher B, et al. Journal of Vascular Nursing. 2017 Sep 1;35(3):146–56.
- Gwilym BL, et al. European Journal of Vascular and Endovascular Surgery. 2021 Jan 7.
- Smith+Nephew 2018. Internal Report. DS.18.260.R.
- Stryja J, et al. Prolekare. 2015;94(8):322 - 328.
- Smith+Nephew 2015. Internal Report. ST865 CT09/02.
- Hudson DA, et al. Int Wound J. 2015;12(2):195-201.
- Payne C, et al. ePlasty. 2014:152-166.
- Malmsjö M, et al. ePlasty. 2014;14:1 - 15.
- Casey C. Consistent delivery of therapeutic negative pressure levels by a single use negative pressure wound therapy system (sNPWT)* in a wound model. Paper presented at: EWMA; 2019; Gothenburg, Sweden.
- Smith+Nephew 2019. Internal Report. RD/19/006.
- Smith+Nephew 2020. Internal Report. 2001002.
- Smith+Nephew 2018. Internal Report. RD/18/132.
- Smith+Nephew 2018. Internal Report. DS/18/219/R V2.
- Mcmanus H, et al. Bacterial retention within a multi-layered absorbent AIRLOCK™ Technology Single Use Negative Pressure Wound Therapy (sNPWT) dressing. Paper presented at: EWMA; 2018; Krakow, Poland.
- Loveluck J, et al. ePlasty. 2016;16:183-195.
- Kilpadi DV, et al. Wound Repair Regen. 2011;19(5):588-596.
- Birke-Sorensen H, et al.. Journal of plastic, reconstructive and aesthetic surgery: JPRAS 64 Suppl, S1–S16 (2011)
- Scalise A, et al. Int Wound J. 2016;13:1260–1281.
- Shim HS, et al. Biomed Res Int. 2018;2018
- Fleming CA, et al. J Hosp Infect. 2018;99(1):75–80.
- Gillespie BM, et al. Surg Innov. 2015;22(5):488–495.
- Galiano RD, et al. PlastReconstr Surg Glob Open. 2018;6(1):e1560.
- Pellino G, et al. Surg Innov. 2014;21(2):204–212.
- Pellino G, et al. Int J Surg. 2014;12 Suppl 2:S64–S68.
- Selvaggi F, et al. Surg Technol Int. 2014;24:83–89.
- Ma Z, et al. Exp Ther Med. 2016;11(4):1307-1317.
- Xia CY, et al. Mol Med Rep. 2014;9(5):1749-1754.
- Groenen H, et al. eClinicalMedicine (part of The Lancet group). 2023;62:102105.
- Bullough L, et al. The Clinical Services Journal. 2015:2-6.
- Hyldig N, et al. BJOG. 2019;126(5):619-627.
- Karlakki SL, et al. Bone Joint Res. 2016;5(8):328-337.
- Nherera LM, et al. Wound Repair Regen. 2017;25(3):474-482.
- Saunders C, et al. BJS Open. 2021;0(0):1 - 8.
- Holt R, et al. British Journal of Hospital Medicine. 2015;76(4). 43.
- O’Leary DP, et al. Ann Surg. 2017;265(6):1082–1086.
- Hudson DA, et al. International wound journal, 2015; 12(2), pp.195-201.
- Witt-Majchrzak A, et al. Pol PrzeglChir. 2015;86(10):456-465.
- Fleming CA, et al. J Hosp Infect. 2018; 99:75–80.
- Smith+Nephew 2016. Internal report. DS.16.179.R.
- Smith+Nephew 2016. Internal report. DS.16.174.R.
- Smith+Nephew 2008. Internal report. DS/08/062/R1.
- Smith+Nephew 2009. Internal report. DS/08/078/R2.
- Smith+Nephew 2008. Internal report. DS/08/062/R2.
- Smith+Nephew 2017. Internal report. DS/16/363/R2.
- Smith+Nephew 2019. Internal Report. EO.AWM.PCSgen.001.v2.